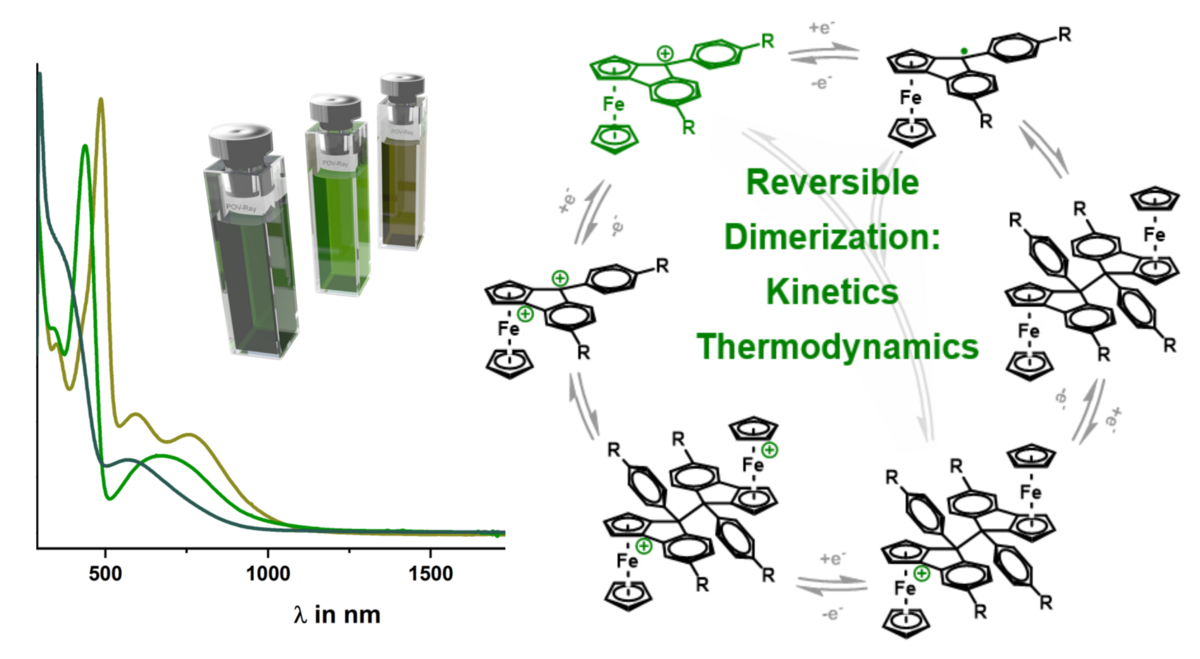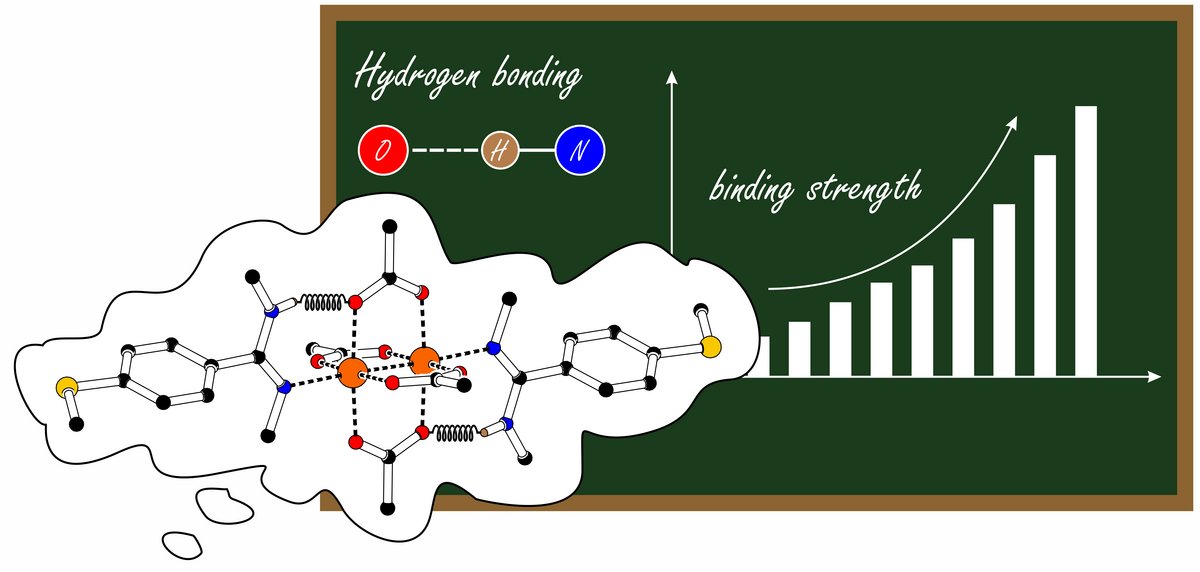
André's Paper in ZAAC is one of the most read papers in ZAAC
André's Paper "Synthesis and crystal structures of rhodium acetate paddle-wheel complexes with anchor group-functionalised and hydrogen bond-supported axial ligands" appeared in August of 2022. Almost two years later, it belongs to ZAAC's most downloaded papers
Weiterlesen